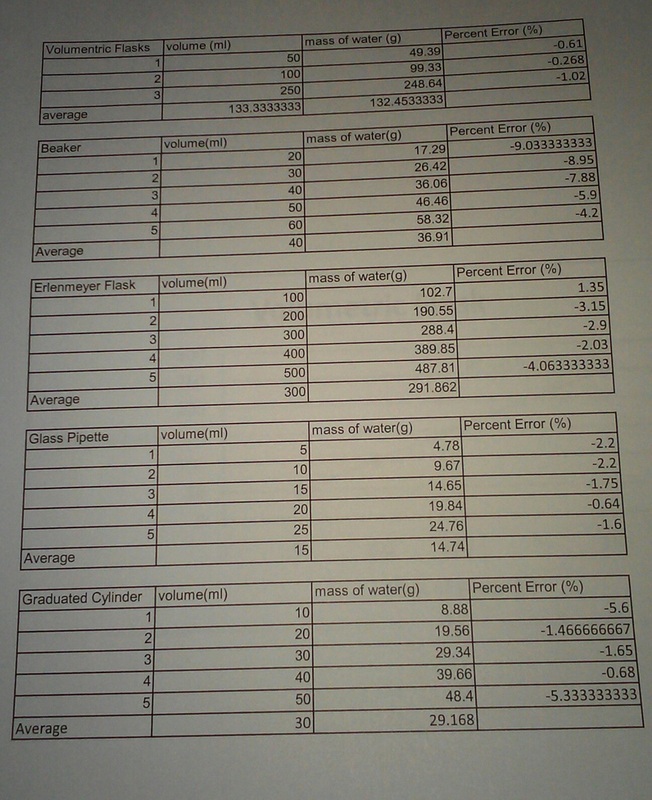
| Introduction: The purpose of this lab was to learn how to use various lab equipment, practice measuring accurately and precisely, and finally cooperating with my fellow team members to finish this experiment. I chose this lab not only because of the points, but also to have experience with measuring and handling lab equipment so I can perform better in future experiments. I was hoping to find which instrument out of the 5 my group and I chose measured most accurately. Two essential components you need to know in order to proceed with this lab are precision and accuracy. Accuracy represents the correctness, or closeness one value is to the true value, where precision represents how many times you can repeat your accuracy or how close the measurements are to each other. Once you understand these concepts, you'll have a better understanding to perform this experiment. Conclusions and Discussions: Percentage Error: When measuring, my group found that the most accurate piece of equipment was the Volumetric flask because when we compared the data, the utensil with the lowest percentage error was the Volumetric flask. Although it was neck to neck with the graduated cylinder, the Volumetric flask turned out to be the most accurate equipment to use in this project. R Squared Value: Once again the ideal choice for measurement would be the Volumetric flask. The reason I believe the Volumetric flask was the most precise is because when I analyzed the graphs, the R squared value of the Volumetric flask was the biggest number, or the closest to the next measurement. Therefore, it would be the most precise instrument to measure with. Best Measurement: I believe that the Volumetric flask should be the ideal measuring tool because when I analyzed the data and answered the previous questions, the Volumetric flask seems to be the most precise instrument. Therefore, it should be the best tool to use because it gets the best results compared to the other utensils. Lab Performance: I think my group did an overall exceptional job through out this experiment. We received guidance from Amanda and did the measurements as precise as we could. When measuring the water we even poured the water until all of the drops came out. However, no matter how precise we try to be, there's always errors that cannot be controlled. So I think what could've contributed to errors in our performance would be the fact that we're human and can't be precise. |
|
0 Comments
Leave a Reply. |
BettyMy name is Betty and this is my chemistry blog where I'll perform all sorts of cool science experiments. Enjoy! Archives
May 2015
Categories |
 RSS Feed
RSS Feed
